
I am not sure of how correct my knowledge is. This is the way I personally see it at least. So they would still be more active going to the left-hand side of the table. As you will learn in class, this leads to smoothly varying trends in properties such as ionization energy and atomic radius as one moves both down the periodic. Second, assuming they could do both (lose and gain) it would still be far easier to lose 2 or 3 electrons than add like 6 more to fill the valance shell. Introduction: The structure of the periodic table is such that elements with similar properties are aligned vertically in columns called groups. Now intuitively it could be thought that an atom would like to have an octet of valance electrons and since most of the nonmetals are on the right-hand side of the table, the closest "octet" would be to the right, achieved by gaining electrons, and the closer it is to the right, the more it wants that electron (becomes more active like halogens).īut for metals it's the very opposite, they want to LOSE electrons because Nonmetals can do both, lose and gain electrons. Well first off metals can ONLY lose electrons, so they will always either gain a positive charge or will have a positive oxidation state. A screenshot is preferable to a picture of your laptop screen. Intro Trends in the Periodic Table Reactivity Stile Education 10.3K subscribers Subscribe Subscribed 2. contains non-metal close non-metal Element that is a poor conductor. Please do not ask for help acquiring, preparing, or handling illicit substances or for help with any activity that does not fall within the confines of whatever laws apply to your particular location.īonus points: If submitting a picture please make sure that it is clear. Group 7 close group 7 The vertical column of non-metal elements next to group 0 in the periodic table, also called the halogens. Any infractions will be met with a temporary ban at the first instance and a permanent ban if there is another. It is also important that you describe the specific part of the problem you are struggling with. It is OK if you are a little (or a lot!) stuck, we just want to see that you have made an effort. The Royal Society of Chemistrys interactive periodic table features history, alchemy, podcasts, videos, and data trends across the periodic table. Please complete any questions as much as you can before posting. We will not do your homework for you, so don't ask. When an electron is added to such a small atom, increased electron–electron repulsions tend to destabilize the anion.Please flair yourself and read over the rules below before posting. In contrast to the chemistry of the second-period elements, the chemistry of the third-period elements is more representative of the chemistry of the respective group.ĭue to their small radii, second-period elements have electron affinities that are less negative than would be predicted from general periodic trends. The anomalous chemistry of second-period elements results from three important characteristics: small radii, energetically unavailable d orbitals, and a tendency to form pi (π) bonds with other atoms. Consequently, the elements of the third period (n = 3: Na, Mg, Al, Si, P, S, and Cl) are generally more representative of the group to which they belong. The chemistry of the second-period element of each group (n = 2: Li, Be, B, C, N, O, and F) differs in many important respects from that of the heavier members, or congeners, of the group. The lack of chemical reactivity implied by these failures was of significance in the development of theories of atomic structure. In fact, all late 19th- and early 20th-century efforts to prepare chemical compounds of argon failed. 
Unique Chemistry of the Lightest Elements The repeating properties in each row of the periodic table, as observed by Mendeleev and others, reflect the repeating electron configurations in subsequent rows. This result was significant because fluorine is the most reactive element in the periodic table. The semimetals lie along the diagonal line separating the metals from the nonmetals and exhibit intermediate properties. Consequently, the elements in the upper right of the periodic table are the smallest and most electronegative the elements in the bottom left are the largest and least electronegative. In contrast, atomic size decreases from left to right and from bottom to top. 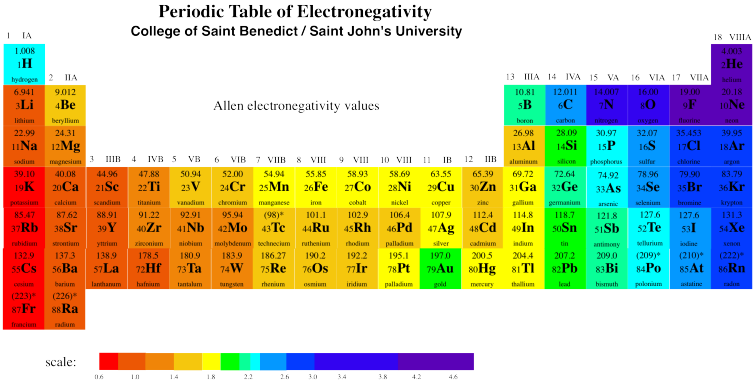
Ionization energies, the magnitude of electron affinities, and electronegativities generally increase from left to right and from bottom to top. Key groups include alkali metals, alkaline earth. The table is divided into metals, nonmetals, and metalloids, each with distinct properties. 
Elements in the same group share similar characteristics, like reactivity. \): Summary of Periodic Trends in Atomic Properties. The periodic table organizes elements into groups and periods based on their chemical and physical properties.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
